Valve material ranges from soft material such as rubber or plastic to exotic valves made from titanium. Of all the materials, the primary material type is metal, with the most common metals being steel and brass. Despite their popularities, many people do not fully understand where steel comes from. To better appreciate the different types of steel and brass, it is critical to start the investigation from the ground up, literally.
From Rock to Iron
Despite its popularity, Steel is not a naturally occuring material. Instead, steel is formed by alloying, or combining , of other naturally occurring elements, mainly, Iron (Fe) and Carbon (C). Thanks to the law of entropy, Iron is found in nature in various oxidized forms such as Magnetite (Fe3O4), Hematite (Fe2O3), Deothite (FeO(OH)), Limonite, or Siderite., typically intertwined inside rocks. When the concentration of these iron oxide is high enough, such as above 50% of the rock’s mass, we refer to them as iron ores and these rocks becomes the raw ingredient of iron.
For reference, the main source of raw iron in USA comes from hematite (Fe2O3), which accounts for more than 3/4 of total US production and is mainly mined from open pits in the Lake Superior region.

First refinement process: Ore to Iron
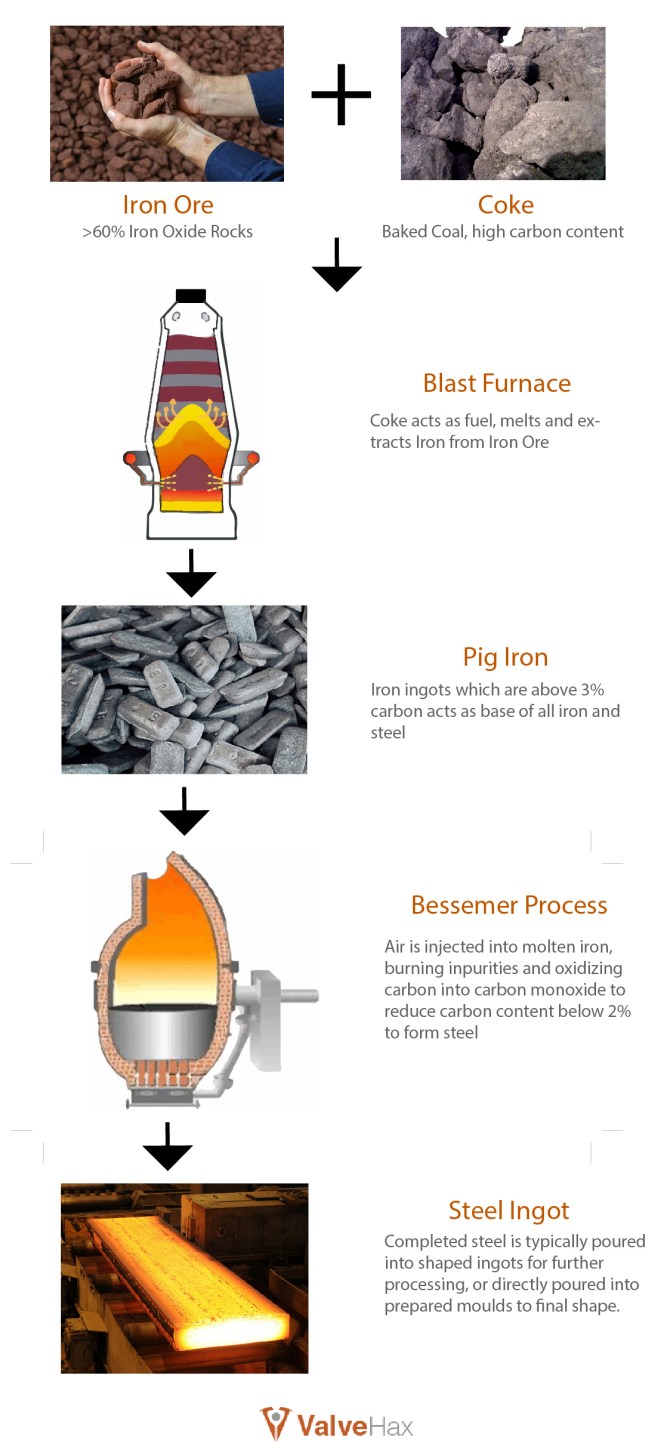
As the best iron ore is only about 60 to 65% iron content, they must be processed to usable form of iron. To accomplish this, iron ore is placed into Blast furnaces with a source of fuel, typically Charcoal or Coke, and melted down to extract the iron from the ores and remove impurities. The charcoal and coke used to melt the iron from the ores is one of the primary source of carbon for the first stage of iron production.
The molten metal is pour into pieces of ingots called PIG IRON; so named for the resemblance to suckling piglets with the way the pig irons was traditionally lined up to the main runner. However, modern day pouring of pig iron typically use automation and revolving patterns, so this sight of many pig irons lined up a runner is no longer common.

After the pig iron is formed, they are not quite ready to be used for final production. As the pig irons usually have a relative high amount of carbon (more than 3%) and have other various contaminating elements such as silicon, manganese, phosphorus and sulfur which need to be further refined. These uncontrolled contaminants causes the iron to be brittle and cannot be worked as-is from the blast furnace. Thus, the pig iron requires further processing with other additives and processes before being capable of used for the final product.
Second refinement process: Decarburization
The second process of refinement is the removal of impurities and removal of carbon. The importance of carbon removal may be strange without a basic understanding of characteristic of Iron-Carbon alloys. One such tool to help breaking down how a molten pool of iron will turn out is the Iron-carbon alloy eutectic diagram, which shows what kind of microstructures the final solid will have depending on the different percentage of carbon in the alloy mixture and heat treatment.

The carbon percentage can be found on the X axis, starting with 0% and ends at 6.67%. Observing carefully, one can see that steel is iron-carbon mixture below 2.06% Carbon, with any alloy with higher carbon content in the realm of iron. Recall that pig iron have a carbon content of more than 3%. To turn pig iron into ingredient for steel making, and most cast iron, requires additional extraction of carbon from the metal.
If we think of this like cooking, we know that adding to a mixture is easy, but taking something out is a rather difficult task. Thankfully, human was able to create method of extracting carbon as early as the Han Dynasty, done very slowly through oxidation of the carbon while stirring the molten metal. This method slowly evolved to potting and stamping. Advancement in this process led to puddling method in the 19th century, ushering in the Industrial Revolution. However, what really introduced modern day steel usage is the introduction of the Bessemer process.
The Bessemer Process
The modern iron decarburization method was formalized by an English gentlemen named Henry Bessemer in 1856. The Bessemer Process works by introducing air to the molten metal and allow it to burn off the impurities and carbon. The oxygen in the air binds with carbon in the molten metal to form carbon monoxide and carbon dioxide, in turn, reducing the carbon content of the melt and turns iron into steel. This process does not require any additional fuel, as the very impurities and carbons within the molten metal acts as the necessary fuel for heat.
As what used to take days to accomplish can now be done in minutes, this process revolutionized steel making and allow steel price to be lowered, leading to the eventual proliferation of steel. While this process was very powerful, it could only remove carbon and silicon, but not phosphorus. This challenge was later overcome by the usage of limestone. When limestone is used to line the converters instead of the traditional clay, it acts as a flux which help draws out the phosphorus to form slag and is known as “basic” Bessemer process. Where the traditional bessemer process was known as “acid” Bessemer process.
In the modern steel making industry, the idea of the bessemer process is retained and upgraded to what is called Basic Oxygen Steelmaking/Linz-Donawitz-Steelmaking. The new process replaces the injection of air with injection of low pressurized (150 psig) pure oxygen. This advancement further reduce the time, effort and cost of steel making as well as increased the purity of the produced steel. For comparison sake, traditional open hearth process may take 12 hours where’s the oxygen process would take less than 1 hour to accomplish the same goal, with better result.
Steel Making Summary
The details of where steel comes from is very broadly generalized here and is put into a simple flowchart below. However, at every stage of this steel making process there are continual advancement and variations. Some example are advance processes which pre-refine the iron ores before it is turned into pig iron. Pig iron also comes in multiple grades depending on the different starting ore and fuel used. Finally, the refinement and extraction of impurities can be changed to captured method to make the process more sustainable. All these are interesting topics which can be a seperate articles alone.
It is important at this point to note that most modern steel making, such as the making of the valve uses scraps as the main source of raw material, melted using electric arc furnaces or Electric Induction furnaces, instead of working from iron ore or pig irons.